After a vaccine has been made and before it can be injected into a persons arm, it needs to be transported. Many vaccine are extremely sensitive to temperature and as a result, keeping the so-called cold chain intact is absolutely critical in the logistics process.
A cold chain is a system for distributing products (such as meat and chemicals) at low temperatures. Cold chains can be separated into three main types:
- Refrigerated cold chains (between 2 and 8 degrees Celsius)
- Frozen cold chain (around -20 degrees Celsius)
- Deep freeze cold chains (around -70 degrees Celsius)
Distributing vaccines internationally is a complicated process and according to one study, 5 to 20 percent of other vaccines, in general, spoil during distribution.
The Case of the COVID Vaccines
The COVID vaccines are especially sensitive to temperatures. According to the Scientific American Magazine “Moderna’s vaccine has to be shipped at –20 degrees Celsius (–4 degrees Fahrenheit), and it can then be stored at that temperature for six months. Once thawed and kept in a refrigerator between two and eight degrees C (36 to 46 degrees F) it is good for up to 30 days. Pfizer’s vaccine must be kept at –70 degrees C (–94 degrees F)—a much greater challenge. Once transferred to a refrigerator, it must be administered within five days.”
Cold Chain Monitoring
Designing a cold chain is one thing. Making sure the cold chain is successful is another. Therefore, quality control is imperative. The process of monitoring cold chains has become increasingly more sophisticated in recent years, with the advent of real-time monitoring solutions that utilize powerful sensors for data logging and to create alerts.
In terms of complexity the deep freeze cold chain required for Pfizer’s mRNA-based Pfizer-BioNTech COVID-19 vaccine’s global distribution is particularly interesting due to the critical environment requirements of the vaccine. It is therefore worth taking a closer look at the cold chain monitoring solution being used in its distribution process.
Controlant Supply Chain Monitoring
Controlant, a young company that specializes in real-time supply chain visibility solutions, has been assigned the role of monitoring Pfizer’s BioNTech Covid-19 vaccine. This was announced in a recent press release by the company.
In it, Tanya Alcorn, Vice President, Biopharma Global Supply Chain for Pfizer is quoted saying “Controlant’s reusable, real-time data loggers and visibility and analysis platform integrates Pfizer’s existing control tower technologies to help manage temperature proactively, identify and react expeditiously to any events that can impact the supply chain, all while automating quality and logistics processes.”
According to Gisli Herjolfsson, Co-founder and CEO of Controlant, the company is also working directly with the U.S. Government and health officials, such as the Center for Disease Control (CDC), U.S. Department of Health and Human Services (HHS), and stakeholders in Operation Warp Speed, to provide on-site monitoring at U.S. Government-preferred designated vaccination locations to help seamlessly ensure continuity during hand-off, actionable communication, and visibility, throughout the entirety of the U.S. supply chain journey of the vaccines.
IoT + Visibility Solutions
The strength of the Controlant monitoring technology is the combination of powerful screen-based IoT tags with sensors, that link to GSM networks and then feed into web and mobile apps. This offers a combination of monitoring reliability and a user friendly interface .
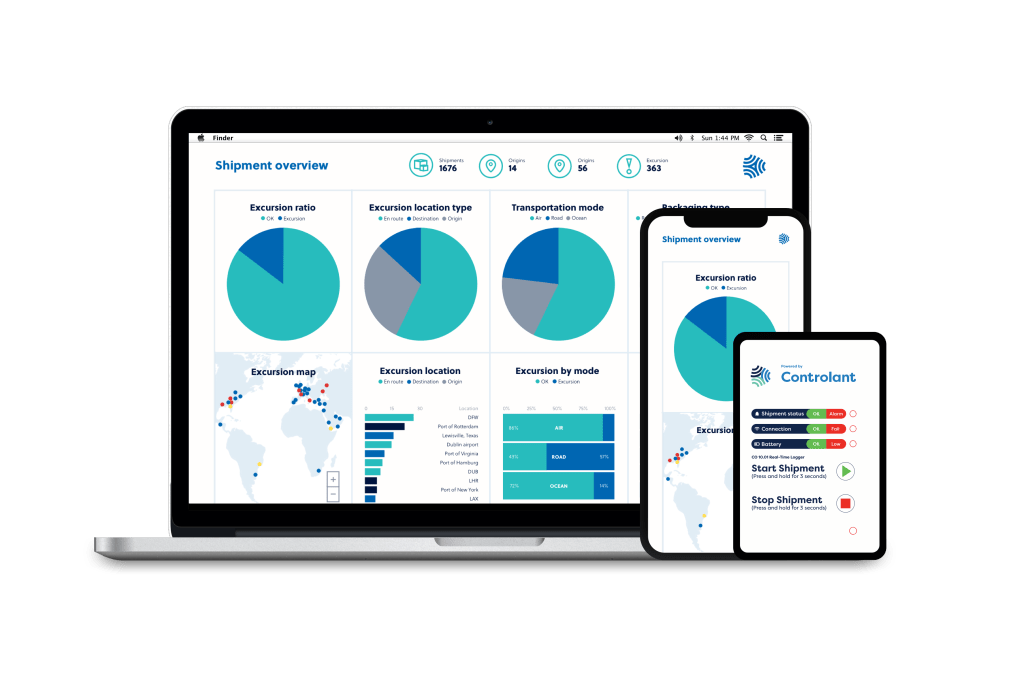
The sensors in Controlant’s IoT devices capture a variety of environmental information, such as temperature and light events. The information is sent in real-time to Controlant’s data logging and monitoring platform, allowing the user to monitor the status of the cold chain in real-time.
